 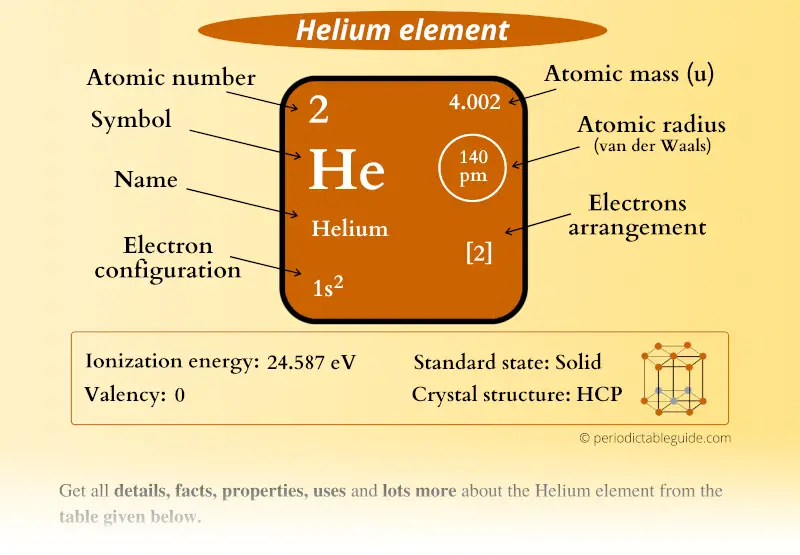
This quantum-mechanical effect is sometimes referred to as due to the exchange force it has no classical analog. The electrons act as if they were subject to a force that depends on the relative orientation of the spins. This results in a smaller binding energy for the singlet states. The net result is that in the singlet state the electrons are in general close together and the Coulomb repulsion is larger. The binding energy for the second electron will be smaller than that for the first electron because the first electron tends to shield the nucleus and thus reduce the effective Coloumb field seen by the second electron.ġ s 2 s 1 S 0 1 s 2 s 3 S 1 ( 1 s ) ( 2 p ) 1 P 1 ( 1 s ) ( 2 p ) 3 P 0, 3 P 1, 3 P 2įor the singlet states, the spin function is antisymmetric under the interchange of the two electrons and the orbital wave function is symmetric for the triplet states, the spin wave function is symmetric under the interchange of the two electrons and the orbital wave function is antisymmetric. The z component of the total spin angular momentum, which is the sum of the z components of the spin angular momenta of the two electrons, will be zero it can be shown that the total spin angular momentum will also be zero. Thus, the quantum numbers ( n, l, m l, m s) for the first electron will be (1, 0, 0, 1 2) and the quantum numbers for the second electron will be (1, 0, 0, − 1 2). Since the spin has little effect upon the energy levels, a lower energy is obtained by putting the second electron in the same orbital state as the first electron but with a different value for the projection of the spin angular momentum along the z axis. Since the second electron cannot be in the same quantum state and thus have the same quantum numbers as the first electron, it must be either in a different orbital state or in a different spin state. Journal reference: Science, DOI: 10.1126/science.To obtain the energy levels for He one adds a second electron to the He + ion. Millard Alexander of the University of Maryland in College Park calls it “very sexy nuclear chemistry”. Most reactions involve far too many particles for this to be practical, but Truhlar says the hydrogen reaction was just simple enough. The way any physical system changes with time can, in theory, be predicted from the quantum states of its particles. 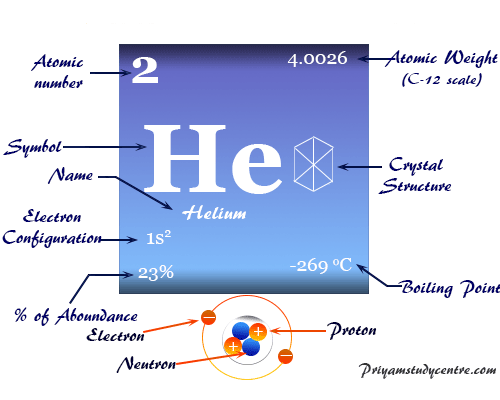
The rates perfectly matched predictions from quantum mechanical calculations led by Fleming’s teammate Donald Truhlar of the University of Minnesota in Minneapolis. ‘Very sexy’Īs expected, the reaction with the disguised helium was the slowest, followed by normal hydrogen, then the light hydrogen. Chemists formed this by replacing the proton in a hydrogen atom with an antimuon, the muon’s positively charged antimatter partner. The team compared how long this took with the rate of the same reaction using normal hydrogen, and with a reaction rate recorded in 1987 when a type of ultra-light hydrogen, called muonium, was used. The helium atoms captured the muons, then pulled hydrogen atoms away from the molecular hydrogen and bonded with them. The difference is that the nucleus is 4.1 times heavier than normal.įleming’s team shot muons produced at the TRIUMF accelerator in Vancouver into a cloud of helium, molecular hydrogen and ammonia. The remaining electron then behaves as if it were orbiting a nucleus with just one positive charge, just like the electron in a hydrogen atom. Donald Fleming of the University of British Columbia in Vancouver, Canada, and colleagues managed to disguise a helium atom as a hydrogen atom by replacing one of its orbiting electrons with a muon, which is far heavier than an electron.īecause it is so heavy, the muon sits 200 times closer to the helium nucleus than the electron it replaces and cancels out one of the nucleus’s positive charges. A hydrogen atom has just one proton and one electron. 
The camouflaged atom behaves chemically like hydrogen, but has four times the mass of normal hydrogen, allowing predictions for how atomic mass affects reaction rates to be put to the test.Ī helium atom consists of a nucleus containing two positively charged protons and two neutrons, encircled by two orbiting electrons which carry a negative charge. 
In a feat of modern-day alchemy, atom tinkerers have fooled hydrogen atoms into accepting a helium atom as one of their own. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
